Different Concentrations of Ammonium Sulfate on Daphnia
Introduction
Using ammonium sulfate(NH4)2SO4 to test Daphnia is a way to test to toxicity in aquatic wildlife since they are at the bottom of the aquatic food chain. If it affects the Daphnia, it will affect the rest of the wildlife in that ecosystem. Ammonium sulfate is tested in lakes, ponds, other water resources because they are composed in plant fertilizers. That can cause runoff from agriculture into an aquatic ecosystem and can harm the organisms. The problem of the use of ammonium sulfate is affected world wide. According to wiseGEEK, “it can cause a range of undesirable plant conditions, such as excessive leaf growth, weak plant bodies, and excessive thatch in lawns. Several countries have also instituted a ban on this form of fertilizer due to its use as an explosives component by terrorist groups.” Ammonium sulfate can also cause many problems to humans including a fatal result. “Will cause irritation in the gastrointestinal tract like nausea, vomiting, and diarrhea, although it isn’t toxic unless consumed in large quantities. Contact with the skin or eyes will cause irritation, redness, itching, and pain. It may also be a neurotoxin, meaning it can cause confusion and behavioral changes,” stated aluminumsulfate.net/ . Does toxicity of the environment for affect the living conditions of all living aquatic organisms. Will the concentration of ammonium sulfate affect all Daphnia? I think that all Daphnia in every solution will die because it is exposed to a new environment that it is not used to. The only solution that the Daphnia will survive is the 0.00% because it is used to living in that type of environment where it isn't toxic to them. |
Methods and Materials |
A. What was done?
Five different solutions of ammonium sulfate were prepared for five per Daphnia in one solution because we wanted to know the toxicity that they can survive in. Depending on the concentration of ammonium sulfate in each solution, we recorded the mortality rate of the number of Daphnia introduced and the number of Daphnia that were dead after 24 hours. B. How was it done? There were five different concentration of ammonium sulfate(0.00%, 0.02%, 0.03%, 0.04%, 0.05%). There were five trials that we did for the whole class. Five Daphnia were introduced into each solution and after 24 hours, we recorded each solution in every trial. C. What was used to do it? We prepared 70 mL of ammonium sulfate solutions of 0.00%, 0.02%, 0.03%, 0.04%, 0.05%. We then fished out five Daphnia for each solution that was raised in spring water. There was five trials with five different concentration of ammonium sulfate with five Daphnia per solution. |
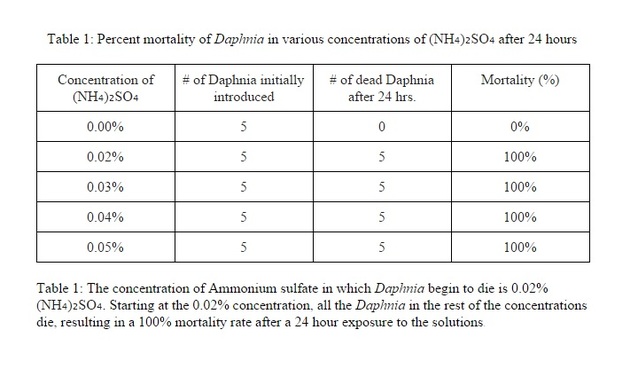
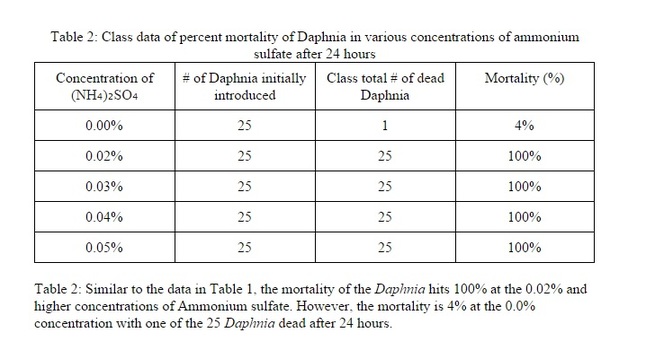
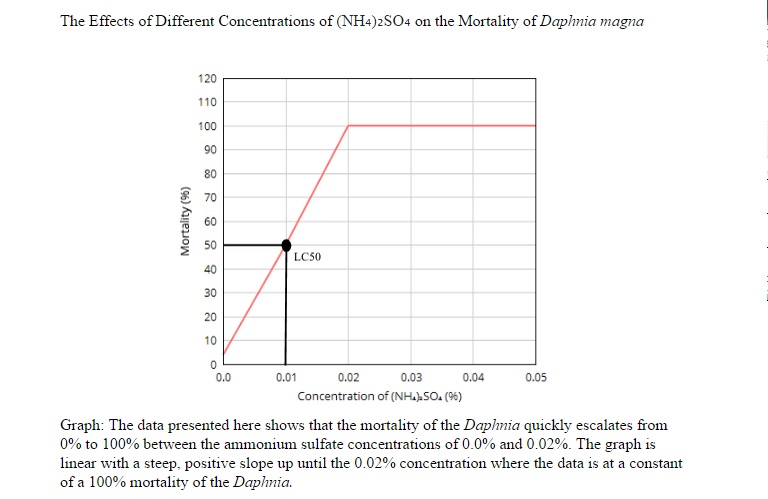
Results
Conclusion
The toxicity of the environment for Daphnia does have an effect on them. The higher amount of ammonium sulfate on the solutions, the higher the mortality rate. Most of the Daphnia had died amongst the solutions that are higher than 0.02%. Although almost all of the Daphnia that were in the control variable lived, there was one that had died for an unknown reason i.e. old age, transferring from tank to solution, etc. The data does support my hypothesis because almost all of the Daphnia in solutions higher than 0.02% had died. The reason that they died could have been the pH of the solutions had changed drastically from their original living source. Another reason that they might have died is that the Daphnia could have been asphyxiated when they were in the solution during the 24 hour period or it could've been under chronic stress from the different living environment.