Bubbles ProtocolIntroduction:
Carbon dioxide and the ocean mix very well because the ocean just soaks in million metrics of CO2. This maintains the the pH of the ocean's water and it helps marine organisms build their shells, bones, and photosynthesis for marine plants. Carbon is absorbed to bond with the water molecules such as hydrogen to form carbonic acid which then its separated into hydrogen ions and bicarbonates. This results in the increase of acidity of the ocean which reduces the amount of coral reefs around the body of waters. This relates to the this protocol because it mimics the gas exchange and pH changes between Earth's atmosphere(blowing CO2 into the beaker) and oceans. Hypothesis: I think that blowing carbon dioxide into the saltwater will cause the pH to increase Methods and Materials: Click pictures for information Conclusion:
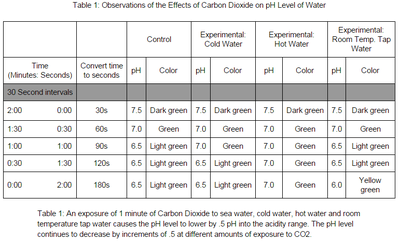
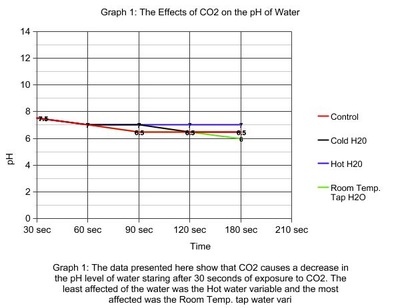
The control variable/seawater decreases after 1 minute to a pH of 6.5 and remains constant throughout the experiment. The cold water variable decreased at a much slower rate with the same pH of 6.5. The hot water didn't change as much remaining at a neutral pH of 7 and decreased by a pH of 0.5. The room temperature tap water continuously decreased to a pH of 6.0 which made it more acidic than the others. Carbon dioxide has an affect on the acidity of the ocean because the ocean is just a carbon sink. |
Shells ProtocolIntroduction:
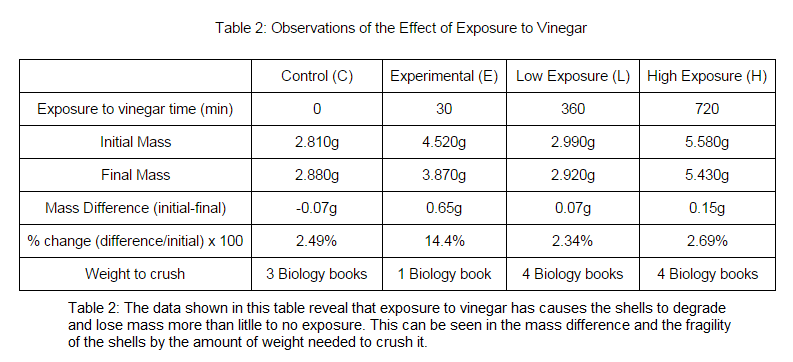
Ocean acidity affects/dissolve the calcium carbonate skeletons and shells of the marine organisms even if it's just a slight change to the pH level. This causes carbonate ions to be scarce. This affects shell fish especially because it causes their shells to become thinner and more vulnerable to break resulting for predators to eat them easily. This relates to the protocol because a low pH (0.0-6.0) or the acidity of the ocean will cause the shells of shell fish to become weakened and fragile. Vinegar and saltwater that was close to a neutral pH (7.0) was used to test if a more acidic solution will disintegrate the shell, resulting in a lighter mass than before. Hypothesis: I hypothesize that a decrease in the pH of seawater on calcium carbonate shells will increase the growth of the shells. Methods and Materials: Two untreated shells were used for this procedure. One for a control variable labeled "C" and the other labeled "E" for the experiment variable. Both shells were massed to record the difference from before putting them into the solutions. The solutions consisted of one beaker with 150 mL of vinegar for the experiment variable and 150 mL of saltwater for the control variable. The untreated shells were then put into these solutions for a 24 hour period and then were taken out to be massed again which has obviously decreased before it was tested. To test the strength of the shells after being put into the solutions, textbooks were put on top of the shells until it cracked or got crushed into pieces. |
Conclusion:
The more acidic the solution was, the more likely the shell was going to break and be more fragile. Therefore, the more acidic the ocean is, the more likely it will disintegrate calcium carbonate skeletons, shells, and coral reefs. This is harmful to the aquatic ecosystem because it damages shelter for the organisms and protection from predators.
The more acidic the solution was, the more likely the shell was going to break and be more fragile. Therefore, the more acidic the ocean is, the more likely it will disintegrate calcium carbonate skeletons, shells, and coral reefs. This is harmful to the aquatic ecosystem because it damages shelter for the organisms and protection from predators.